European Pharmacopeia (EP), British Pharmacopeia (BP) methods for Antibiotic Potency Testing
The OMNICON EP/BP Zones of Inhibition software performs all calculations in accordance to the procedures defined in the EP / BP Methods, sometime referred to as the 3+3 Method. The software allows the Analyst to specify a minimum of 4 plates and maximum of 10 plates per assay, defines the concentration of the standards and preparations, and the assumed potency.
Once the zone diameters are measured, all the calculations are performed and the data presented in the "Independent Assay" report. The Analyst also has the ability to generate a "Combination Analysis" report by selecting Independent assay previously analyzed.
Plate Reading
The OMNICON Zones Reader reads each 100 mm petri dish with 6 zones in less than 3/10th of a second, measuring the diameter and shape of the zone. The shape value is used to exclude zones which are oval due to spills or irregular growth.
21 CFR, Part 11, Electronic Records and Signatures
The OMNICON EP/BP Zones of Inhibition software is compliant, providing three levels of security, audit trails and data encryption.
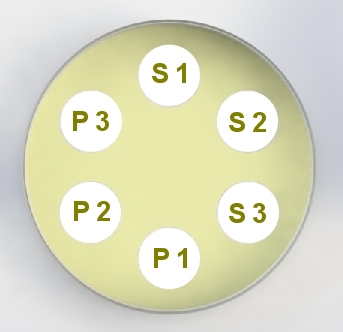
Assay Layout
|
6 zones per plate Replicates |
References
United States Pharmacopeia (USP)
US-FDA Code of Federal Regulations (CFR)
Association of Analytical Communities (AOAC International)
British Pharmacopeia (BP)
European Pharmacopoeia (EP)
Society of Japanese Pharmacopeia (JP)
|
|
|
|
OMNICON is a registered trademark of BioLogics, Inc